Trinity provides risk-based validation services that support equipment, process, and facility performance across the full lifecycle.
We deliver practical, risk-based validation services that align equipment, processes, and people across the full lifecycle. From early planning through execution and requalification, our teams help you achieve defensible compliance without slowing your business.
Validation should prove that systems perform as intended, under real conditions, for their actual use. Trinity approaches validation as an operational discipline, not a paperwork exercise.
Our services are grounded in regulatory expectations, risk-based thinking, and hands-on execution. We focus on clarity of scope, defensible rationale, and documentation that stands up during inspections and over time.
End-to-end CQV support aligned with FDA, EU GMP, and industry guidance. We help define scope early, execute IQ/OQ/PQ efficiently, and ensure deliverables reflect how systems are actually used.
Beyond equipment, we support a full range of validation activities that ensure your processes remain consistent, reproducible, and compliant over time, giving you confidence in reliability and regulatory readiness.
We design and execute temperature mapping studies that demonstrate uniformity, stability, and control for critical storage and process environments.
We support data integrity and validation activities that ensure systems, records, and reports are complete, accurate, and reliable throughout their lifecycle.
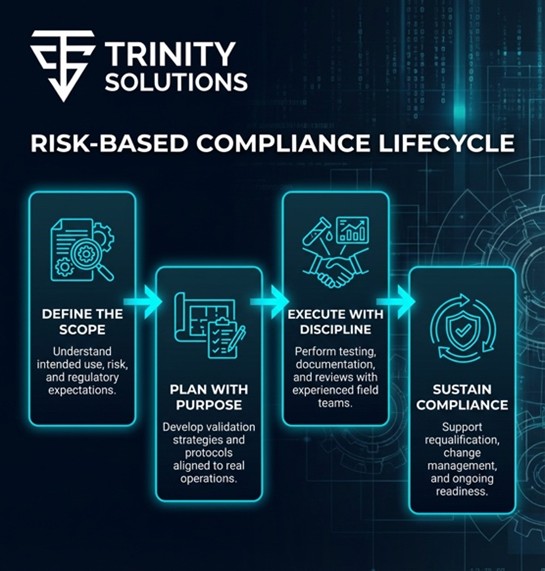
Our approach is grounded in how regulated systems are actually designed, operated, and reviewed. Rather than applying one-size-fits-all validation, we focus on intended use, system criticality, and risk to patient, product, and data. This allows validation activities to be scaled appropriately, executed consistently, and defended with confidence during regulatory review.
Across each phase of the lifecycle, we align planning, execution, and documentation to real operational conditions. Validation strategies are developed early, execution standards are applied consistently in the field, and results are evaluated with an emphasis on traceability and impact. Ongoing support ensures changes are assessed properly, systems remain in a validated state, and compliance is sustained over time.
Our validation methodologies align with global regulatory expectations and industry best practices.
Standards Supported
Whether you need targeted validation support or full lifecycle execution, Trinity brings clarity, structure, and experience to your validation programs.